Frequently Asked Questions: Antigen Testing in Schools
Antigen tests use a quick analysis to detect active virus in individuals, returning results within 15 minutes. In schools, antigen tests can be used on-site, sometimes referred to as “point-of-care” for the entire asymptomatic population without known exposure to detect previously unidentified positive individuals. If an individual does test positive, the school can quickly isolate that person, quarantine close contacts, and thereby reduce the risk of viral transmission. In a school setting, antigen testing is used more as a screening tool to identify potentially positive individuals, not as a diagnostic tool as it might be used in healthcare settings.
Modeling studies have demonstrated that point-of-care or self-administered surveillance tests with fast turnaround time or frequent testing have high epidemiological value and can significantly mitigate transmission of cases in schools. These high-frequency tests of asymptomatic populations hold significant promise in stopping the spread of disease. For more information on antigen testing, please see: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-guidelines.html.
COVID coordinators/lead school nurse or school/district administrators can contact Susan Haberstroh at susan.haberstroh@delaware.gov, charter schools can contact Kendall Massett at kendallm@decharternetwork.org, and private/parochial schools can contact Dana Carr at dana.carr@delaware.gov to initiate. DDOE has also developed https://www.doe.k12.de.us/Page/4243 to assist districts, charters, private and parochial schools with implementation.
DPH will provide the test materials, which may include an analyzer (for BD Veritor test) or test cards (for Abbott BinaxNOW). As of December 2020, DPH has sufficient supplies for antigen testing until at least through the 2020-2021 school year. DPH will work with schools to determine the best approach, supplies and equipment that might be needed, facilitate scheduling, and address any outstanding questions.
For the first two weeks of the testing, DPH will serve as the ordering physician.
DPH recommends that schools/districts identify a community-based ordering provider (MD/DO, PA, NP) to provide any necessary guidance or assistance related to interpreting tests or other medical matters. This individual might be associated with a local medical practice or health care system associated with a school-based wellness center (if applicable), or be a medical provider with an association to the school from the community.
Please contact Dana Carr at dana.carr@delaware.gov for assistance in identifying a sponsoring volunteer healthcare partner if the district/school is unable to do so.
DPH will work with districts/schools to identify and provide the optimal number of tests/analyzers on an ongoing basis. Factors to consider include the total number of students to be tested, cadence or frequency of testing, or available support for oversight.
For the BD Veritor analyzer, DPH recommends 1 unit per 1,000 students to begin. Analyzers can be shared between school buildings or between individual schools (i.e. three small charter schools may collaborate and share the test analyzer between them).
For those using the Abbott BinaxNOW tests, DPH will work with the district or school to determine supply needs commensurate with the operational testing plan.
The lead school nurse/COVID coordinator will work with DPH to determine an adequate supply of tests based on the testing plan. DPH will provide a two-week supply of tests at a time. As this initial supply is depleted, the districts/schools will re-order additional supplies using the SHOC Resource Request Form, at which time they will indicate their ordering provider for continued testing.
The lead school nurse or an assigned individual can serve as the primary point of contact for a district or school, with support from school-level nurses as needed, for the day-to-day testing logistics and protocols. The actual testing administration, management of materials from classes, collection of samples, etc. may be overseen by the school nurse, if time allows, or other staff and/or volunteers may provide support and oversight to students in collecting samples and completing the testing analysis. Older students are likely able to effectively manage their own sample collection and management.
Online training is available and recommended for anyone supporting testing efforts.
For the Abbott BinaxNow test, training and information is available at: https://www.globalpointofcare.abbott/en/support/product-installation-training/navica-brand/navica-binaxnow-ag-training.html
For BD Veritor test, training and information is available at: https://www.bdveritor.com/training/
Test sites will need to apply for a waiver from The Centers for Medicare & Medicaid Services (CMS) Clinical Laboratory Improvement Amendments (CLIA), which regulates the quality that all laboratories must meet. For an initial applicant, the CLIA Initial Application must be completed so that testing sites can manage the accuracy, reliability, and timeliness of patient test results.
For more information on obtaining a CLIA waiver and the form to complete, please see: https://www.cms.gov/files/document/cms-clia-laboratory-quick-start-guide-remediated.pdf.
Yes, participating students must have consent from a parent/guardian or if over 18 years old, can provide consent. Sample consent forms are available upon request from Susan Haberstroh at susan.haberstroh@delaware.gov. They can be signed and collected in paper format or electronically using a system such as DocuSign or similar.
SARS-CoV-2 antigen tests are generally less sensitive than the tests given in the community setting, which are nucleic acid viral tests that use reverse transcription polymerase chain reaction (RT-PCR). Although antigen tests are considered to be highly sensitive and specific to detecting active virus, cases of false positives have been reported.
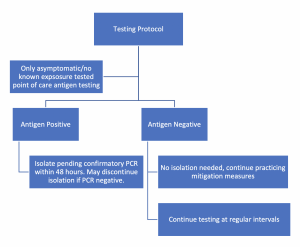
Because of the risk of false positives, the Delaware Division of Public Health (DPH) recommends using the antigen testing as a screening tool, with confirmatory PCR testing within 48 hours of the positive antigen test.
Testing Procedures
The test is a nasal swab, which may be administered to a student by an adult; an older student may administer to themselves independently.
For more information on administering the BD Veritor test, please see: https://www.bdveritor.com/wordpress/wp-content/uploads/2020/08/BD-Veritor-System-for-Rapid-Detection-of-SARS-Co-V-2-Instructions-for-Use.pdf.
For more information on administering the Abbott BinaxNOW, please see: https://www.globalpointofcare.abbott/en/support/product-installation-training/navica-brand/navica-binaxnow-ag-training.html.
Staff administering tests or handling samples should wear a medical facemask and gloves. Gloves should be disposed of as regular garbage and do not need to be included as biohazardous waste.
Staff may want to have a stopwatch (separate from a phone), pens, name labels for samples, a trash can, and information sheets ready for the testing day.
For the BD Veritor, test cartridges develop for 15 minutes before being placed into an analyzer, which outputs a reading of positive or negative.
For the Abbott BinaxNOW, test cards develop for 15 minutes and read directly—two lines indicates a positive result, one line a negative result.
The primary purpose of the testing is for asymptomatic students. Symptomatic students should be immediately isolated and sent home from school pending clearance by a licensed medical professional.
At this time, all K-12 teachers have access to routine, periodic testing through Vault. DPH recommends that these tests be used primarily for students.
This will depend on the makeup of the school, the number of students learning in person or participating in extra-curricular activities. Depending on the number of students, districts/schools may prioritize populations by grade level (focusing on secondary students, for example), student groups (such as students with special needs who cannot wear face coverings or remain socially distant), or individuals who interact with others more (such as sports teams or students who see multiple support staff).
DPH recommends students be tested once per month, potentially on a rotating basis. Districts/schools should develop an operational plan for testing before beginning.
As the process is adopted and becomes more streamlined in the school setting, up to 100 tests/day can be reasonably expected.
Results are submitted for each rapid, point-of-care antigen test performed for COVID-19 to the Division of Public Health (DPH) within 24 hours by a dedicated and HIPAA-compliant web-based DPH portal.
According to the Delaware’s Department of Natural Resources and Environmental Control (DNREC), schools generating waste as a result of on-site rapid antigen testing for the COVID-19 virus will need to manage all waste from suspected or confirmed COVID-19 patient specimens and kit components as infectious waste. More information about the specific items considered to be infectious waste, instructions on disposal, and other additional materials can be found at https://www.doe.k12.de.us/Page/4243.
If the student tests positive after an antigen test, the student would be isolated and sent home. The student should seek a confirmatory PCR test in the community to occur within 48 hours. The student should remain in isolation until the test results are confirmed. If the PCR test is POSITIVE, the student is considered a confirmed positive case and the school nurse should undertake the typical contact tracing process.
If the student has a NEGATIVE PCR result taken within the 48 hours the antigen test is considered a false positive and the student can stop isolation.
Stats on COVID-19
For more data on Delaware COVID-19 cases including demographic breakdowns, go to My Healthy Community



